탄소 나노튜브(CNT) 탄소나노튜브(CNT)는 대표적인 1차원 나노물질로서, 뛰어난 기계적 강도(강철보다 100배 높음), 탁월한 전도성, 우수한 열적 특성, 그리고 독특한 광학적 특성 덕분에 에너지 저장, 복합재료, 생체의학, 전자 기기 등 다양한 분야에서 응용 가능성이 매우 높은 물질로 주목받고 있습니다. 그러나 CNT 사이의 강한 반데르발스 힘(~500 eV/µm)과 높은 종횡비(>1000)로 인해 CNT는 쉽게 응집되어 우수한 성능을 발휘하고 실제 응용 분야에 적용하는 데 심각한 제약을 받고 있습니다. 따라서 용매 또는 고분자 매트릭스 내에서 CNT를 균일하고 안정적으로 분산시키는 것은 CNT의 나노 규모 특성을 최대한 활용하고 대규모 응용을 촉진하는 데 필수적인 전제 조건입니다. CNT의 응집은 주로 넓은 비표면적, 튜브 벽 사이의 강한 반데르발스 힘, 그리고 sp2 혼성화된 탄소 원자에 의해 형성된 비편재화된 π 전자 구름 사이의 π-π 스태킹 상호작용 때문입니다. 이러한 응집 현상은 비표면적을 감소시킬 뿐만 아니라 매트릭스 내에서 연속적인 전도성 또는 보강 네트워크 형성을 저해합니다. 현재까지 탄소나노튜브(CNT) 분산을 위한 두 가지 주요 방법, 즉 공유 결합 기능화와 비공유 결합 기능화가 개발되었습니다. 공유 결합 기능화는 가용성 기능기 또는 친수성 사슬을 CNT에 접합함으로써 CNT의 분산성을 크게 향상시킬 수 있습니다. 비공유 결합 기능화는 첨가된 분산제를 이용하여 비공유 상호작용(반데르발스 힘, 수소 결합, 소수성 상호작용, 정전기적 인력 등)을 통해 CNT 측벽에 흡착시킴으로써 이루어집니다.

현재 탄소나노튜브(CNT) 측벽의 간접적 및 직접적 화학적 기능화를 위한 두 가지 방법이 개발되었습니다. 간접적 방법은 일반적으로 화학 반응을 통해 CNT 표면에 활성 부위를 생성하는 것입니다. 가장 대표적인 예는 강산을 사용하여 CNT를 산화시켜 -COOH, -CHO, -OH와 같은 산소 함유 기능기를 표면에 생성하는 것입니다. CNT의 분산성을 더욱 향상시키기 위해 추가적인 아민화 또는 아실화 반응을 통해 CNT 측벽을 개질할 수 있습니다. 그림 1에서 볼 수 있듯이, 산화된 CNT는 산-염기 반응을 통해 옥타데실아민(CH3(CH2)17NH2)과 직접 결합하여 양쪽성 이온을 형성하거나, 티오닐 클로라이드(SOCl2) 또는 옥살릴 클로라이드((COCl)2)와 반응하여 아실 클로라이드 중간체를 형성한 후, 4-테트라데실아닐린(CH3(CH2)13C6H4NH2)으로 아실화됩니다. 반응 후, CNT 표면에 부착된 긴 알킬 사슬은 용해제 역할을 하여 대부분의 유기 용매에 우수한 분산성을 부여합니다.
다양한 종류의 장쇄 알킬기를 접합함으로써, 탄소나노튜브(CNT)의 다양한 용매에서의 분산성을 효과적으로 조절하여 다양한 기능적 요구 사항을 충족시킬 수 있다. 예를 들어, 수용성 고분자인 폴리(아미노벤젠술폰산)(PABS)을 접합하면 단일벽 탄소나노튜브(SWCNT)의 수용액 분산성을 효과적으로 향상시킬 수 있으며, 생성된 SWCNT-PABS는 순수 PABS보다 훨씬 높은 전도도를 나타낸다. 아실 클로라이드를 이용하여 활성화된 CNT에 글루코사민(C6H13NO5)을 접합하면 분산성이 우수한(0.1-0.3 mg/mL) 기능화된 CNT를 얻을 수 있다. 다중벽 탄소나노튜브(MWCNT)를 강산(H2SO2/HNO2)으로 처리한 후 아미노트리에틸렌글리콜 사슬을 접합하여 양전하를 도입하면 분산성이 우수한 기능화된 MWCNT를 얻을 수 있다.
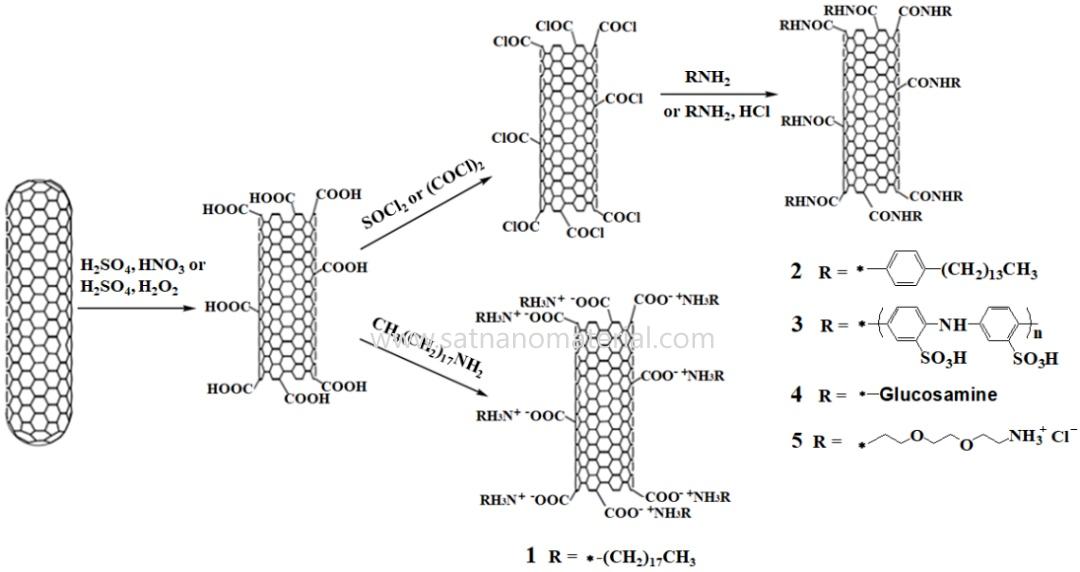
그림 1. 아미드화 반응을 통한 탄소나노튜브의 공유 결합 기능화
에스테르화 반응은 탄소나노튜브(CNT)의 공유 결합 기능화를 위한 또 다른 효과적인 방법입니다(그림 2). 에스테르화 반응을 통해 합성된 도데실 4차 암모늄 브로마이드로 기능화된 단일벽 탄소나노튜브(SWCNT)(그림 2의 6)는 pH 6.87~11.25 범위에서 우수한 수분산성을 나타내며, 폴리비닐알코올(PVA) 기반 복합재료의 충전재로 사용됩니다. 또한, 폴리에틸렌 글리콜(그림 2의 7), PVA(그림 2의 8), DNA 및 단백질과 같은 고분자 세그먼트를 에스테르화 또는 아미드화 반응을 통해 CNT 표면에 공유 결합시켜 수용액에서의 분산성을 향상시킬 수 있습니다.
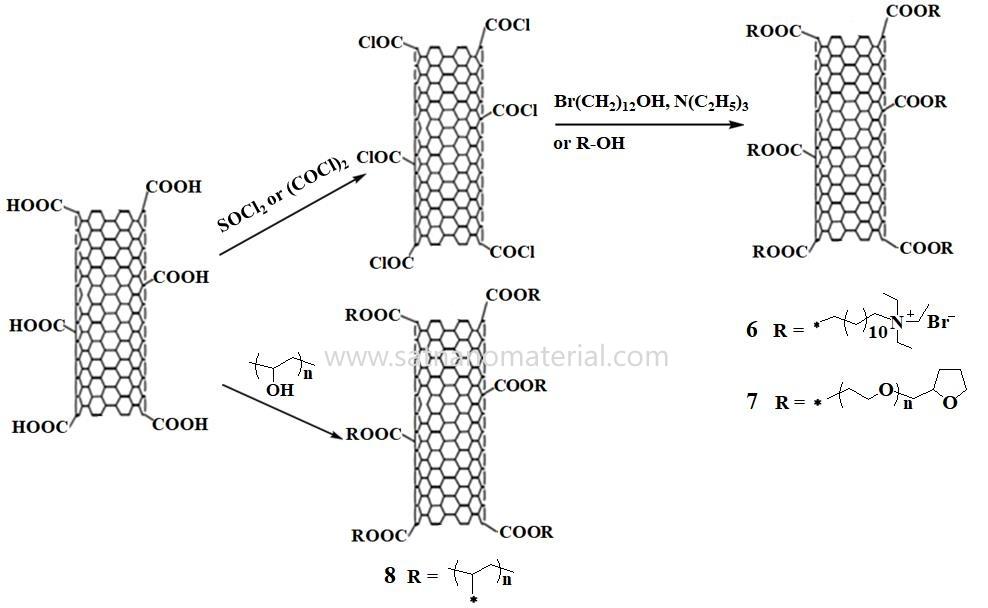
그림 2: 지질화 반응을 통한 CNT의 공유 결합 기능화.
하이드록실기와 실메톡시기 사이의 축합 반응은 탄소나노튜브(CNT)의 화학적 기능화에도 사용되어 왔다. 하이드록실화된 CNT와 폴리(3-트리메톡시실릴프로필 메타크릴레이트)(CCB-PMPS)가 접합된 전도성 카본 블랙의 반응을 통해 테트라하이드로퓨란(THF)에 우수한 분산성을 갖는 CNT 기반 하이브리드 필러가 생성되었다.
앞서 언급한 간접적인 개질 방법 외에도, 탄소나노튜브(CNT) 측벽의 직접 기능화에 대한 연구가 활발히 진행되고 있습니다. CNT는 질소 알켄, 카벤, 이민 일라이드 또는 자유 라디칼(또는 고리화 첨가 반응)과 반응할 수 있습니다. 간접 기능화와 비교했을 때, 직접 기능화는 강산이나 산화 공정으로 인한 CNT 손상을 방지하고 CNT 길이 단축을 막을 수 있습니다. 다음 그림은 단일벽 탄소나노튜브(SWCNT) 측벽의 직접 기능화에 대한 개략도입니다. SWCNT는 질소 알켄, 친핵성 카벤 및 퍼플루오로알킬기와 각각 첨가 반응을 일으킵니다. 알킬 아지드 에스테르 및 비피리딘 이미다졸리딘과 반응하여 얻은 SWCNT는 디메틸 설폭사이드(DMSO)에서 우수한 분산성을 나타내는 것으로 확인되었습니다. SWCNT는 아릴, 수지상 고분자, 긴 알킬 사슬 및 올리고폴리에틸렌 글리콜 단위와 같은 더 복잡한 치환기를 가진 질소 함유 화합물과 반응하며 1,1,2,2-테트라클로로에탄(TCE), DMSO 및 1,2-디클로로벤젠(1,2-DCB)을 포함한 다양한 유기 용매에서 우수한 분산성을 나타냅니다.
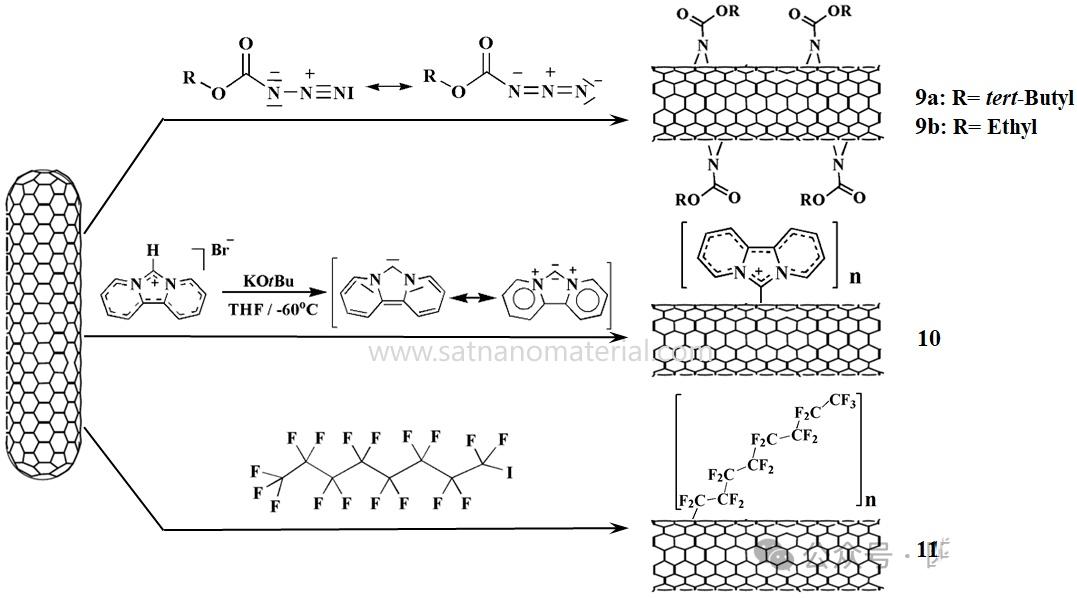
그림 3은 질소 알켄, 친핵성 카벤 및 자유 라디칼의 첨가를 통해 CNT의 측벽을 기능화하는 것을 보여줍니다.
알파 아미노산과 알데하이드의 열 축합으로 생성된 질소 함유 메틸 알칼리 일라이드의 1,3-쌍극자 고리화 첨가 반응은 탄소 나노튜브(CNT)를 기능화하는 효과적인 방법임이 입증되었습니다. 페놀기는 1,3-쌍극자 고리화 첨가 반응을 통해 단일벽 탄소 나노튜브(SWCNT) 표면에 접합될 수 있으며, 극성 용매에서 안정적인 분산성을 나타냅니다. 알데하이드와 변형된 글리신을 접합에 사용하면 염화칼륨(CHCl₃), 염화수소(CH₂Cl₂), 아세톤, 메탄올, 에탄올, 물과 같은 용매에 분산될 수 있는 생성물을 얻을 수 있습니다. 또한, tert-부톡시카르보닐(Boc)로 보호된 아미노 말단기를 갖는 N-기능화 글리신과 1,3-쌍극자 고리화 첨가 반응을 통해 아민 기능화 및 수분산성 유도체로 CNT를 기능화할 수 있었습니다(그림 4: CNT 측벽에 대한 1,3-쌍극자 고리화 첨가 반응 기반 CNT 기능화). 이 공정은 이후 생의학 분야에 적용하기 위해 아미노산 및 펩타이드로 변형된 탄소 나노튜브를 제조하는 데 사용되었습니다.
위에서 설명한 탄소 나노튜브의 공유 결합 변형 및 분산 기술 공유는 SAT NANO의 기술자인 다나가 SAT NANO의 탄소 나노튜브 분말을 분산시키는 과정을 통해 완료했습니다. 이 기술 공유가 공유 결합 변형 분산 탄소 나노튜브의 분산에 도움이 되기를 바랍니다. 다음 글에서는 다나가 비공유 결합 변형 분산 탄소 나노튜브의 분산 기술을 소개할 예정입니다.
SAT NANO는 최고의 공급업체입니다. 탄소 나노튜브 분말 아르 자형 중국에서 문의사항이 있으시면 언제든지 저희에게 연락주세요. admin@satnano.com